Immunosome®-Caproylamine (Non-PEGylated)
SKU# IMS-2052
Description
Numerous techniques have been developed to prepare immunoliposomes based on the nucleophilic reactivity of free amine groups of proteins or peptides. One of the most popular and commonly used methods is to covalently couple free carboxylic groups to primary amines through activation of the carboxyl groups with EDC (1-ethyl-3-[3-dimethylaminopropyl] carbodiimide). EDC, which is a so-called zero-length crosslinking agent, reacts with the carboxyl to form an amine reactive intermediate (O-acylisourea). The produced O-acylisourea can be easily displaced by nucleophilic attack from primary amino groups in the reaction mixture. However, this intermediate is unstable and hydrolyzed in aqueous solutions. In order to prevent the intermediate hydrolysis, sulfo-NHS (N-hydroxysulfosuccinimide) is added to EDC to produce a significantly more stable and more soluble active intermediate (NHS ester).
Consequently, the immunoliposomes are prepared by a two-step coupling procedure: first, activating the free carboxyl group on the antibody, peptide or protein with EDC and sulfo-NHS, and then covalently conjugating antibody to the lipids through displacement of sulfo-NHS groups by amine groups of the liposomes, as depicted below. EDC/sulfo-NHS coupling reactions are highly selective and highly efficient, and the biological activity of the protein or peptide is preserved.
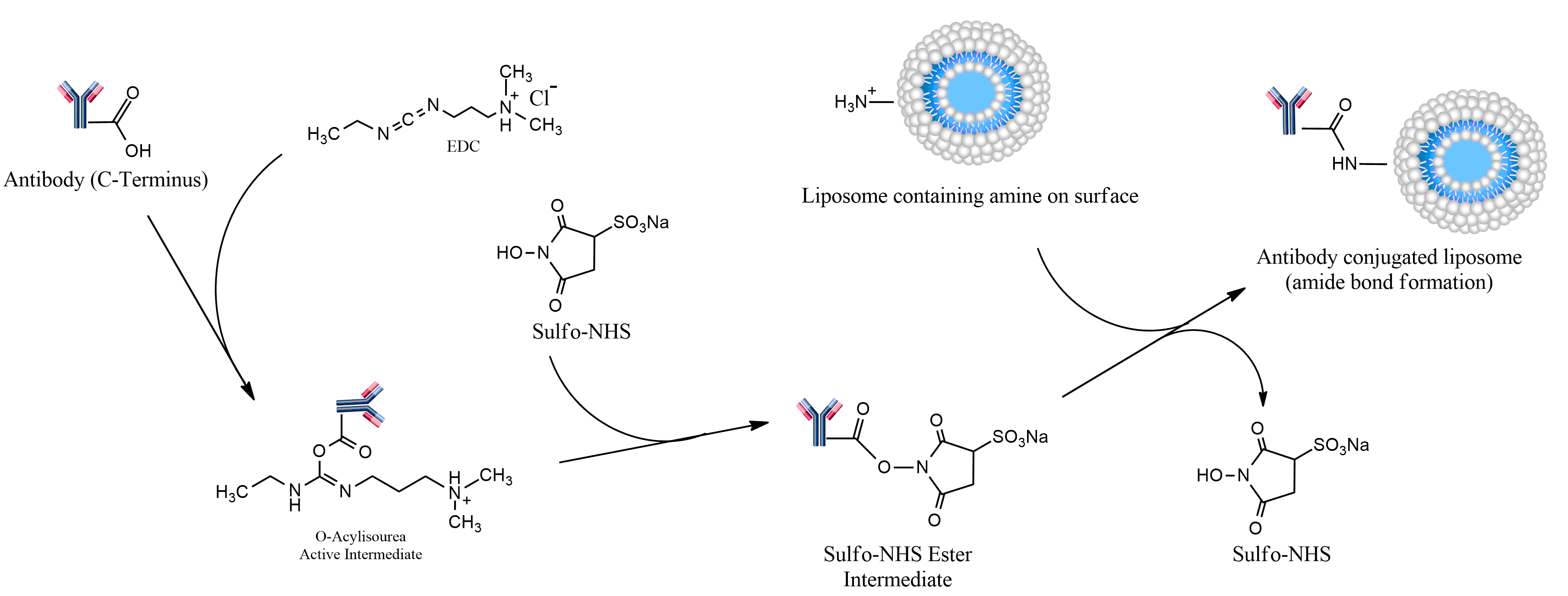
Immunosome®-Caproylamine is a non-PEGylated product. For other carboxylic acid reactive (PEGylated and non-PEGyalated products) and also Immunosome® products suitable for other types conjugation methods see here.
Formulation Information
Immunosome®-Caproylamine (Non-PEGylated)
| Lipid Composition | Concentration (mg/ml) | Concentration (mM) | Molar Ratio Percentage |
|---|---|---|---|
| L-alpha-Phosphatidylcholine | 12 | 15.5 | 69 |
| Cholesterol | 2.6 | 6.73 | 30 |
1,2-Dioleoyl-sn-glycero-3-phosphoethanolamine-N-(hexanoylamine)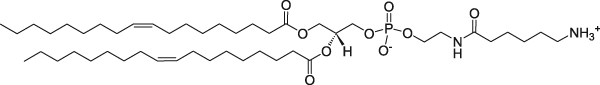 | 0.19 | 0.22 | 1 |
| Total | 14.79 mg/ml | 22.45 mM | 100 |
| Buffer and Liposome Size | Specification |
|---|---|
| Buffer | Phosphate Buffered Saline |
| pH | 7.4 |
| Liposome Size | 100 nm |
Conjugation Protocol
Materials and Equipment
In order to conjugate your antibody, protein, peptide or ligand containing carboxylic acid to Immunosome®-Caproylamine liposomes you will need:
- EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride). The solution should be made fresh moments before use.
- Sulfo-NHS (N-hydroxysulfosuccinimide). The solution should be made fresh moments before use.
- MES Buffer: The reason for using MES buffer is because MES is a non-amine, non-carboxylate buffer, and activation reaction with EDC and Sulfo-NHS is most efficient at pH 4.5-7.2. Alternatively, you can use PBS buffer. But you must make sure that the pH of the buffer is adjusted to 6.00.
- Sephadex® spin column: Sephadex size exclusion spin column can be used for separation of liposomes form free EDC (MW: 191.70). Since EDC is being separated from large liposome particles then any sizes of Sephadex® spin column such as G-10, G-15, G-25, G50 can be used. However, keep in mind that you will lose a large percentage of your liposomes on the spin column. Alternatively, instead of removing the EDC by spin column you can quench it by using 2-mercaptoethanol.
- Float-A-Lyzer® with a proper MWCO that easily allows the cleanup of your liposome conjugated ligand from free and non-conjugated protein/peptide/ligand. You need to make sure that the MWCO is below 1,000,000 dalton. At 1,000,000 dalton, the pore size on the dialysis membrane gets close to 100 nm and therefore your liposomes can be dialyzed out. You cannot use dialysis cassettes blindly. Please understand the technique before using either spin column or dialysis cassette. If you do not use the correct MWCO, you can lose your entire prep. For this protocol, we recommend MWCO of 300,000 dalton.
Preparation Method
A two-step protocol for the activation of proteins, peptides or antibodies with EDC/sulfo-NHS and subsequent conjugation with amine-containing molecules is given below.
- Dissolve the protein, antibody, peptide or ligand to be activated in 0.1 M MES, 0.5 M NaCl, pH 6.0 (reaction buffer) at a concentration between 1-10 mg/ml. For most proteins and antibodies (depending on the MW) this is equal to a concentration of 10-5 to 10-4 molar solution. Please calculate the molarity of your protein, peptide, antibody or ligand.
- Add to the solution in step 1 a quantity of 10-fold molar excess of EDC and 25-fold molar excess of sulfo-NHS. To aid in aliquoting the correct amount of these reagents, they may be quickly dissolved in the MES buffer at a higher concentration, and then a volume immediately pipetted into the protein solution to obtain the proper molar quantities. Mix and react for 15 minutes at room temperature.
- Before adding the liposomes containing amine (Immunosome®-Caproylamine) to activated protein or ligand, you need to make sure that EDC will be removed from the solution. This can either be done by using a proper size spin column or by quenching EDC by 2-mercaptoethanol.
- Add 2-mercaptoethanol to the reaction solution to obtain a final concentration of 20 mM. Mix and incubate for 10 min at room temperature. NOTE: If the protein being activated is sensitive to this level of 2-mercaptoethanol, instead of quenching the reaction chemically, the activation may be terminated by desalting (step 5).
- If the reaction was quenched by the addition of 2-mercaptoethanol, the activated protein may be added directly to the amine-containing liposomes (Immunosome®-Caproylamine) for conjugation. Alternatively, or if no 2-mercaptoethanol was added, the activated protein may be purified from reaction by-products by gel filtration using a desalting spin column such as Sephadex® spin column. The desalting operation should be performed rapidly to minimize hydrolysis and recover as much as active ester functionality as possible.
- After purification, add the activated protein to the Immunosome®-Caproylamine. The molar ratio of reactive caproylamine lipid to protein, peptide or antibody is preferred to be around 10:1. The total lipid concentration in our liposomes is 22.45 mM. 1% mol of the lipid in liposomes contains NH2 group and only half of them are exposed to the outside of the liposomes, which is equal to 0.11 mM of reactive conjugable lipid. For 2 ml volume liposomes, this is equal to 2.20×10-7 mol, and for 5 ml volume liposomes, this is equal to 5.50×10-7 mol of NH2. You need to calculate the total mol of your peptide, protein or ligand in your solution and add 1:10 molar ratio of ligand to lipid. They react for at least 2 h at room temperature.
- Remove non-conjugated antibody, protein, peptide or ligand by dialysis. We prefer dialysis to size exclusion columns. Dialysis is a much slower process but there will be minimum loss of immunoliposomes after the prep is cleaned from non-conjugated protein/peptide/ligand. Spin columns are much faster, but you can easily lose over 50% of the liposomes on the spin column. We recommend using Float-A-Lyzer® dialysis cassette from Spectrum Labs. You need to choose a cassette with proper MWCO depending on the MW of your protein, ligand, antibody or antibody fragment. NOTE: If you decide to use a dialysis cassette you need to make sure that the MWCO is below 1,000,000 dalton. At 1,000,000 dalton, the pore size on the dialysis membrane gets close to 100 nm and therefore your liposomes can be dialyzed out. You cannot use dialysis cassettes and spin columns blindly. They come in various sizes and you need to choose the correct size wisely. Dialyze the immunoliposome solution in 1 liter of PBS at pH 7.4 for 8 hours. Change the dialysis buffer with a fresh 1 liter of PBS and let is dialyze for another 8 hours. After this step, your cleaned up immunoliposome is ready to be used.
Liposome Particle Calculator
Immunosomes are unilamellar liposomes and sized to 100 nm. The molar concentration of liposome is 22.45 mM. By having liposome diameter (nm) and lipid concentration (µM), you can calculate the total number of the lipids in one liposome and the number of the liposomes in one milliliter of the liposome solution. To use the calculator click here.
Technical Notes
- EDC and sulfo-NHS should be prepared immediately and kept at room temperature before use.
- The activation reaction with EDC and Sulfo-NHS is most efficient at pH 4.5-7.2, and EDC reactions are often performed in at pH 4.7-6.0. For this reason, we have formulated the liposomes in PBS buffer and adjusted the pH to 6.
- Reaction of Sulfo-NHS-activated molecules with primary amines is most efficient at pH 7-8, and Sulfo-NHS-ester reactions are usually performed in phosphate-buffered saline (PBS) at pH 7.2-7.5.
- Tris buffer should never be used in any step of the process since it contains amine.
- If you are using a ligand or peptide that is hydrophobic then it is recommended to solubilize it in DMSO or DMF and then add the buffer to it. It is recommended not to use more than 5% volume of DMSO or DMF in the solution. DMF and DMSO are both compatible with liposomes and they are also miscible in water. Other organic solvent such as ethanol and chloroform are not compatible with liposomes and will cause the liposomes to lyse. If you end up using DMSO or DMF then after the conjugation reaction is done, you need to remove DMSO and DMF from the liposomes. In order to do that you need to use a dialysis cassette that is made from REGENERATED CELLULOSE MEMBRANE. NOTE: Not all membranes are compatible with DMF and DMSO. We recommend using a Slide-A-Lyzer™ MINI Dialysis Device with MWCO of 2K made from regenerated cellulose membrane manufactured by ThermoFisher. After DMSO or DMF is removed you can use Float-A-Lyzer® dialysis device for the final step of cleaning up the prep.
- Liposomes should be kept at 4°C and NEVER be frozen.
Database
Direct link to the database page for easy navigation: Immunoliposomes Conjugation Database
Appearance
Immunosome®-Caproylamine is a white translucent liquid made of nano size unilamellar liposomes. Usually due to the small size of liposomes no settling will occur in the bottom of the vial. The liposomes are packaged in an amber vial.
Ordering/Shipping Information
- All liposome based formulations are shipped on blue ice at 4°C in insulated packages using overnight shipping or international express shipping.
- Liposomes should NEVER be frozen. Ice crystals that form in the lipid membrane can rupture the membrane, change the size of the liposomes and cause the encapsulated drug to leak out. Liposomes in liquid form should always be kept in the refrigerator.
- Clients who order from outside of the United States of America are responsible for their government import taxes and customs paperwork. Encapsula NanoSciences is NOT responsible for importation fees to countries outside of the United States of America.
- We strongly encourage the clients in Japan, Korea, Taiwan and China to order via a distributor. Tough customs clearance regulations in these countries will cause delay in custom clearance of these perishable formulations if ordered directly through us. Distributors can easily clear the packages from customs. To see the list of the distributors click here.
- Clients ordering from universities and research institutes in Australia should keep in mind that the liposome formulations are made from synthetic material and the formulations do not require a “permit to import quarantine material”. Liposomes are NOT biological products.
- If you would like your institute’s FedEx or DHL account to be charged for shipping then please provide the account number at the time of ordering.
- Encapsula NanoSciences has no control over delays due to inclement weather or customs clearance delays. You will receive a FedEx or DHL tracking number once your order is confirmed. Contact FedEx or DHL in advance and make sure that the paperwork for customs is done on time. All subsequent shipping inquiries should be directed to Federal Express or DHL.
Storage and Shelf Life
Storage
Immunosome® products should always be stored at in the dark at 4°C, except when brought to room temperature for brief periods prior to animal dosing. DO NOT FREEZE. If the suspension is frozen, the encapsulated drug can be released from the liposomes thus limiting its effectiveness. In addition, the size of the liposomes will also change upon freezing and thawing.
Shelf Life
Immunosome®-Caproylamine is made on daily basis. The batch that is shipped is manufactured on the same day. It is advised to use the products within 4 months of the manufacturing date.
References and background reading
1. Hermanson GT. Bioconjugate techniques. Academic press; 2013 Jul 25.











