Fluorescent Phosphatidylserine (PS)-based ATP Liposomes (ATPsome®)
Description
Adenosine triphosphate (ATP) is the most abundant and primary carrier of the required energy for various functions in cells. Prolonged ischemia, reperfusion, anaerobic metabolism and lactate accumulation can lead to a dramatic decrease of ATP, cell swelling, cell rupture, and finally cell death by necrotic, necroptotic, apoptotic, and autophagic mechanisms. Due to drastic hydrolysis of ATP in vivo by ectoenzymes and poor cellular penetration, the direct delivery of ATP to the ischemic tissues is difficult.
To increase delivery of ATP to the tissues and protect from enzymatic degradation, encapsulation in liposomes has been proposed and demonstrated in various models of ischemia [1,2]. Studies on myocardial [1,3,4], liver [5-8], retina [9] and wound healing [10-12] ischemia have shown the ability of liposomal encapsulated ATP to prevent cell death and tissue dysfunction following ischemic events.
The encapsulation of ATP in liposomes markedly promotes its effectiveness by preventing the hydrolysis by extracellular enzymes, increasing ATP circulation time and enhancing its intracellular penetration. Depending on the type of the cell line and the target organ various types of liposomes with different surface charges such as anionic, cationic and neutral has been studied by various groups. Moreover, ATP encapsulated liposomes has been demonstrated to improve energy state and function of the cold-stored liver [6,7,13].
Formulation Information
Fluorescent Phosphatidylserine (PS)-based ATP Liposomes (ATPsome®)
For more information on the lipid composition of the liposomes mentioned above click here.
| Fluorescent Dye | Excitation/Emission (nm) | Molecular Structure |
|---|---|---|
| 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine-N-(7-nitro-2-1,3-benzoxadiazol-4-yl) (ammonium salt) | 460/535 | 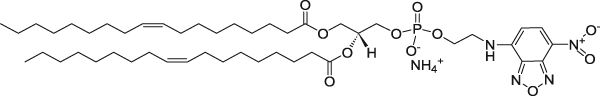 |
| 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine-N-(lissamine rhodamine B sulfonyl) (ammonium salt) | 560/583 | 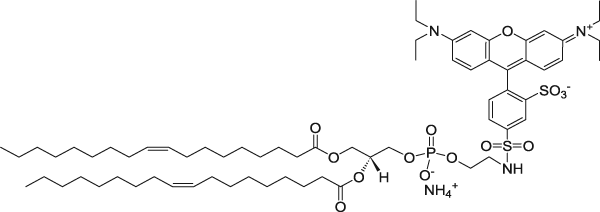 |
| Lipid and Encapsulated ATP | Amount (µmol/vial) |
|---|---|
| Total Lipid | 1.5 |
| Total Encapsulated ATP | 0.5 |
| Buffer, Lyoprotectant and Liposome Size | Specification |
|---|---|
| Buffer | Phosphate Buffered Saline |
| pH | 7.4 |
| Lyoprotectant | Trehalose |
| Liposome size | 100 nm (after hydration) |
Technical Notes
- Due to very rapid hydrolysis of adenosine triphosphate (ATP), ATP liposome (ATPsome®) formulations are all freeze-dried.
- Leftover liposomes in liquid form that are made after hydration of the lyophilized proliposome formulation cannot be used or kept in refrigerator for future use due to rapid hydrolysis on ATP.
- ATP liposome are formed upon hydration of the lyophilized formulation. Very large percentage of ATP molecules will remain encapsulated inside the liposomes, but some percentage will be free and in non-encapsulated form after hydration of the freeze-dried formulation. Free ATP molecules usually do not have any negative impact upon injection of the formulation to the animal or upon adding the formulation to the cell cultures.
- Trehalose is used as a lyoprotectant in all freeze-dried liposome formulations. The size distribution after hydration of the freeze-dried formulation will be around 100 nm.
- Freeze-dried liposomes should be kept at -20°C.
Appearance
Non-fluorescent ATP proliposome (ATPsome®) is a white lyophilized powder. Fluorescent ATPsome® depending on the color of the dye will be slightly colored (see SDS for appearance). Fluorescent ATPsome® containing NBD fluorescent dye is yellow and fluorescent ATPsome® containing rhodamine is pink. The lyophilized formulation is packaged in an amber vial. Liposomes are formed upon adding water to the freeze-dried proliposomes. Non-fluorescent liposomes will form a white translucent liposome formulation and fluorescent liposomes will form colored translucent liposomes depending on the color of the fluorescent dye.
Ordering/Shipping Information
- All proliposome lyophilized formulations are shipped on dry ice at -78.5°C in insulated packages using overnight shipping or international express shipping.
- Lyophilized proliposomes should be frozen at -20°C. The liposomes are formed upon adding water to proliposomes. These liposomes should be used immediately due to very rapid hydrolysis of ATP.
- Clients who order from outside of the United States of America are responsible for their government import taxes and customs paperwork. Encapsula NanoSciences is NOT responsible for importation fees to countries outside of the United States of America.
- We strongly encourage the clients in Japan, Korea, Taiwan and China to order via a distributor. Tough customs clearance regulations in these countries will cause delay in custom clearance of these perishable formulations if ordered directly through us. Distributors can easily clear the packages from customs. To see the list of the distributors click here.
- Clients ordering from universities and research institutes in Australia should keep in mind that the liposome formulations are made from synthetic material and the formulations do not require a “permit to import quarantine material”. Liposomes are NOT biological products.
- If you would like your institute’s FedEx or DHL account to be charged for shipping, then please provide the account number at the time of ordering.
- Encapsula NanoSciences has no control over delays due to inclement weather or customs clearance delays. You will receive a FedEx or DHL tracking number once your order is confirmed. Contact FedEx or DHL in advance and make sure that the paperwork for customs is done on time. All subsequent shipping inquiries should be directed to Federal Express or DHL.
Storage and Shelf Life
Storage
ATP proliposome (ATPsome®) products should always be stored in a freezer at -20°C. The liposomes are formed upon adding water to proliposomes. These liposomes should be used immediately due to very rapid hydrolysis of ATP. The leftover liposomes in liquid form that are not immediately used can be discarded safely in drain.
Shelf Life
ATP proliposomes (ATPsome®) are made to order. The batch that is shipped is manufactured on the same day. It is advised to use the products within 6 months of the manufacturing date.











