Cellsome® made from Diether Lipids
Description
The fundamental structure of cell membranes is bilayers composed of phospholipids, and the vital function of the phospholipids in the membrane is to help keep it fluid and semi-permeable. Conventional glycerophospholipids have acyl chains attached to the sn-1 and sn-2 positions of the glycerol backbone via an ester bond. Ether lipids are a unique class of glycerophospholipids that have an alkyl chain attached to the sn-1 position by an ether bond (glycerol-ether lipids). In ether lipids, the alcohol group attached to the phosphate is generally choline or ethanolamine. Ether-linked phospholipids such as 1-alkyl-2-acyl-phosphatidylcholine and dialkylphosphatidylcholine are also found in the plasma and organelle membranes of mammalian species. Ether lipids form approximately 20% of the total phospholipid in mammals with different tissue distribution; brain, heart, spleen and white blood cells have the highest levels, while liver have a very little amount of ether lipids.
Studies on the formation and thermodynamic properties of ether-linked phospholipid bilayer membranes have indicated that in contrast to ester-linked phospholipid, the formation of the non-bilayer structure takes place spontaneously. This is attributed to the weaker interaction between polar headgroups in the ether-linked than that in the ester-linked phospholipids. It has also shown that the phase behavior of the ether-linked phospholipid bilayer membranes in ambient pressure is almost equivalent to that of the ester-linked phospholipid bilayer membranes under high temperatures and pressures, and the difference in the phase behavior decrease as the alkyl-chain length increases.
Due to distinctive properties of ether lipids, liposomes made from ether lipids exhibit very unique characteristics and performance: a) the ether bonds are more stable than ester linkages over a wide range of acidic or alkaline pH; b) stability properties of the liposomes is enhanced by bipolar lipids, and the saturated alkyl chains gives stability towards degradation in oxidative conditions; c) the unusual stereochemistry of the glycerol backbone enhance the resistance against the attacks by other organism phospholipases.
Phospholipase A2 (PLA2) cannot hydrolyze the ether lipid liposomes. Diether lipids do not go through hydrolysis due to having an ether bond instead of an acyl bond and therefore to do that, they are a suitable candidate for experiments that needs to be performed at a higher temperature for an extended period of time. For more information about hydrolysis and oxidation of phospholipids see here.
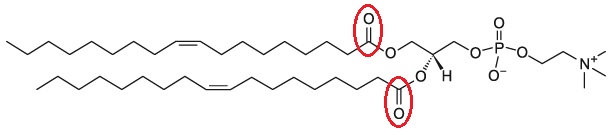
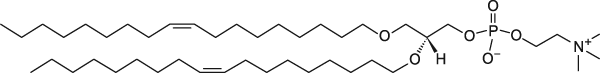
Saturated diether lipids can neither be hydrolyzed nor oxidized.
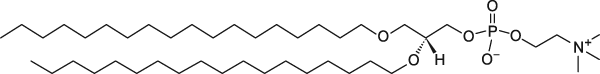
Formulation Information
Cellsome® made from Diether Lipids
For more information on the lipid composition of the liposomes mentioned above click here.
| Buffer and Liposome Size | Specification |
|---|---|
| Buffer | Phosphate Buffered Saline* |
| pH | 7.4 |
| Liposome size | 100 nm |
| * If you prefer different buffers, please specify in your order. | |
Liposome Particle Calculator
Cellsome®-Diether liposomes are unilamellar and sized to 100 nm. The molar concentration of liposome is 10 mM. By having the liposome diameter (nm) and lipid concentration (µM), you can calculate the total number of the lipids in one liposome and number of the liposomes in one milliliter of the liposome solution. To use the calculator click here.
Each 100-nm liposome is composed of almost 80,000 phospholipid molecules. 10 mM liposome solution (phospholipid concentration of 10 mM) has 75.2 trillion (75.2×1012) liposome particles per milliliter of the solution. When using the liposomes calculator, you need to keep two things in mind:
- Enter the concentration in µM. As an example, 10 mM solution is 10,000 µM.
- Only include the concentration of phospholipids in your calculation. Cholesterol does not count since it inserts itself into the hydrophobic fatty acid chains of the lipid.
This calculation only works for the unilamellar liposomes which are below 200 nm in size. It does not work for larger multilamellar liposomes. For more information about the calculation see the video below:
Technical Notes
- Liposomes are unilamellar and sized to 100 nm. If you need them to be made in another size, then you should mention that at the time of ordering. Unilamellar liposomes can be made in 50 nm and 200 nm sizes. Liposomes that are larger than 200 nm are multilamellar structure.
- Liposomes are made in PBS buffer at pH 7.4 but it can be made in any other buffer of your choice. You can specify your buffer at the time of ordering.
- Liposomes are made in degassed buffer that is purged with argon to avoid oxidation of the unsaturated phospholipids.
- Liposomes are made under sterile condition.
- Liposomes are unilamellar and therefore due to their small size they will not settle in the bottom of the vial.
- If you need to take multiple aliquots out of the vial, for use over a period of time, then it is advised to take extreme care in not contaminating the vial. It is recommended to handle the vial under a sterile hood to maintain the sterility of the product.
- Liposomes should never be frozen. Ice crystals that form during freezing will rupture the lipid membrane of the liposomes and change the size the liposomes particles.
- Liposomes should be kept in refrigerator at 4°C in order to avoid the hydrolysis of the liposomes.
Appearance
Cellsome® made from diether lipids is a white translucent liquid made of nano size unilamellar liposomes. Usually due to the small size of liposomes no settling will occur in the bottom of the vial. The liposomes are packaged in an amber vial.
Ordering/Shipping Information
- All liposome based formulations are shipped on blue ice at 4°C in insulated packages using overnight shipping or international express shipping.
- Liposomes should NEVER be frozen. Ice crystals that form in the lipid membrane can rupture the membrane and change the size of the liposomes. Liposomes in liquid form should always be kept in the refrigerator.
- Clients who order from outside of the United States of America are responsible for their government import taxes and customs paperwork. Encapsula NanoSciences is NOT responsible for importation fees to countries outside of the United States of America.
- We strongly encourage the clients in Japan, Korea, Taiwan and China to order via a distributor. Tough customs clearance regulations in these countries will cause delay in custom clearance of these perishable formulations if ordered directly through us. Distributors can easily clear the packages from customs. To see the list of the distributors click here.
- Clients ordering from universities and research institutes in Australia should keep in mind that the liposome formulations are made from synthetic material and the formulations do not require a “permit to import quarantine material”. Liposomes are NOT biological products.
- If you would like your institute’s FedEx or DHL account to be charged for shipping, then please provide the account number at the time of ordering.
- Encapsula NanoSciences has no control over delays due to inclement weather or customs clearance delays. You will receive a FedEx or DHL tracking number once your order is confirmed. Contact FedEx or DHL in advance and make sure that the paperwork for customs is done on time. All subsequent shipping inquiries should be directed to Federal Express or DHL.
Storage and Shelf Life
Storage
Cellsome® products should always be stored at in the dark at 4°C, except when brought to room temperature for brief periods prior to animal dosing. DO NOT FREEZE. If the suspension is frozen, the encapsulated drug can be released from the liposomes thus limiting its effectiveness. In addition, the size of the liposomes will also change upon freezing and thawing.
Shelf Life
Cellsome® made from diether lipids is made on daily basis. The batch that is shipped is manufactured on the same day. It is advised to use the products within 4 months of the manufacturing date.
References and background reading
2. Dean JM, Lodhi IJ. Structural and functional roles of ether lipids. Protein & cell. 2017 May 18:1-1.











